
Imaging of the Pituitary Gland
Authors
Kirsten Forbes, MD
John Karis, MD
William L. White, MD*
Divisions of Neuroradiology and *Neurosurgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
This article reviews the magnetic resonance imaging techniques that can be used to assess the pituitary gland and describes the imaging characteristics of normal sellar anatomy and common sellar and suprasellar pathologies.
Key Words: macroadenomas, magnetic resonance imaging, microadenomas, pituitary
Undoubtedly, magnetic resonance (MR) imaging is the optimal imaging technique for evaluating the pituitary gland. Adequate assessment requires dedicated high-resolution imaging through the sellar region. The coronal plane offers the best single view for assessing the sella and allows the pituitary gland to be distinguished from the surrounding structures. Sagittal views are particularly helpful for evaluating midline structures. Because the pituitary gland is small, high spatial resolution images are required. Slice thicknesses must be 3 mm or less, and the field of view must cover only the sellar and immediate parasellar regions. T1-weighted sequences, with and without gadolinium, are optimal for evaluating both normal anatomy and underlying pathology. The amount of gadolinium used is field-strength dependent with a one-half dose typically being used at 1.5 Tesla. Fat-saturation techniques are useful for postoperative evaluations.[14]
Normal Anatomy
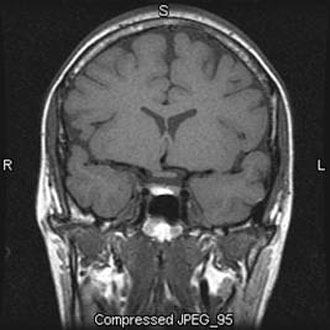
The anterior and posterior lobes of the pituitary can often be differentiated on MR imaging. In adults the anterior lobe is isointense to the remainder of the brain, and the posterior lobe is hyperintense on T1-weighted sequences (Fig. 1).[11] This finding is thought to reflect the high concentration of neurosecretory granules in the neurohypophysis, but the exact substrate is still debated. In neonates and during pregnancy, distinguishing the anterior from the posterior pituitary may be challenging because the anterior lobe also appears hyperintense on T1-weighted images.

Absence of the normal posterior pituitary bright spot should prompt a search for an ectopic posterior pituitary that has failed to migrate inferiorly from the hypothalamus (Fig. 2). This finding can also be associated with transection of the pituitary stalk. However, absence of the pituitary bright spot can be a normal finding or may reflect technical factors. It should not be interpreted as an absolute indication of pathology.
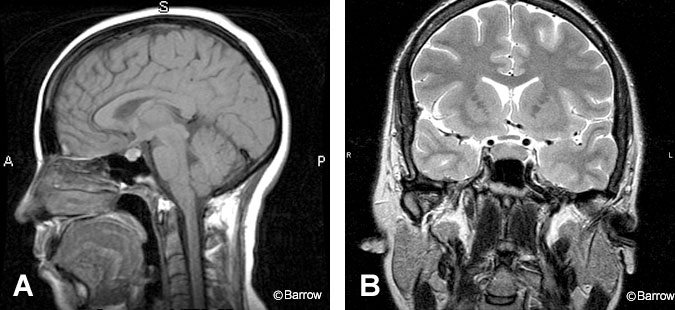
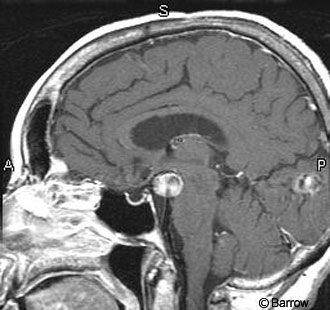
Figure 3. During puberty and pregnancy the pituitary gland undergoes physiological hypertrophy and shows a convex upper margin, as seen on (A) sagittal T1-weighted and (B) coronal T2-weighted images. Figure 4. The empty sella turcica, shown in this sagittal T1- weighted image, is a normal anatomic variant. An incidental basilar artery aneurysm is present.
The size of the pituitary gland varies with physiological status. In the normal state, the gland is less than 8-mm high. During puberty or pregnancy, it becomes larger and more upwardly convex (Fig. 3). During puberty it may reach 10-12 mm in females and exhibits even more marked changes during pregnancy.[9] Chanson et al. report that the pituitary height exceeds 9.0 mm in less than 0.5% of healthy women age 18-35.[3] They report one healthy 24-year-old female with a normal pituitary gland height of 12 mm and width of 12 mm. Another female patient with a normal pituitary gland measured 16 mm in width. They emphasized that physiological pituitary hypertrophy seems to be a frequent cause of incidentaloma and that careful examination of the MRI may help distinguish this entity from pituitary tumors and infiltrating lesions. Physiological pituitary hypertrophy is confirmed by normal baseline pituitary function and extensive hormonal tests. Identification of these patients is important to avoid unnecessary pituitary surgery.[3] It is normal for the pituitary gland to be markedly flattened against the floor of the sella, a configuration known as an empty sella turcica (Fig. 4). The sella largely becomes filled with cerebrospinal fluid (CSF) because of the enlarged diaphragmatic sella hiatus, and it may appear to expand with CSF pulsations.
Pituitary Microadenomas
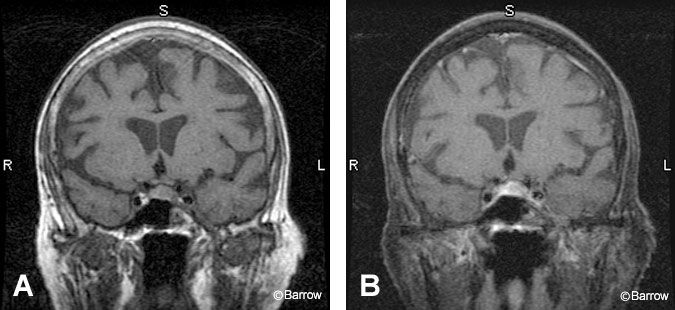
The findings associated with a pituitary microadenoma on MR imaging are often subtle, necessitating a high-quality imaging study (Fig. 5). With optimal MR technique, about 90% of these small tumors (10 mm or less in diameter) can be identified.[14] False-negative studies are most often associated with Cushing’s disease becau se many tumors that secrete adrenocorticotropic-hormone (ACTH) are small. A microadenoma can alter the contour of the pituitary gland, creating an outward convexity of its superior or inferior aspects. Classically, tumors secreting ACTH, thyroid stimulating hormone, luteinizing hormone, and follicle stimulating hormone are found centrally within the pituitary gland while prolactin and growth hormone adenomas occur at the periphery. Laterally placed microadenomas may cause the infundibulum to deviate from the tumor. However, a tilted infundibulum can also be a normal finding observed in about 50% of normal subjects.[1]
Pituitary microadenomas are easier to identify on T1-weighted sequences than on T2-weighted sequences because they are often isointense to normal pituitary tissue on the latter. On T1-weighted sequences, microadenomas tend to appear as focal hypointense lesions. Occasionally, they are isointense or even hyperintense to the surrounding pituitary. Susceptibility artifacts from the interface between the sphenoid sinus and sella may project as low signal foci in the pituitary gland and can mimic a microadenoma.[10] Administration of intravenous gadolinium improves sensitivity, particularly when lesions are isointense on precontrast T1-weighted imaging.[17]
Compared to the normal pituitary gland, microadenomas show delayed gadolinium uptake. They are best detected by imaging immediately after contrast administration when they appear as an area of relative nonenhancement. Some centers use dynamic MR imaging during contrast administration, and this technique may improve sensitivity slightly.[4] The time of maximal signal difference between microadenoma and normal gland is variable although it occurs within a few minutes of contrast administration.[4] Imaging more than 30 minutes after intravenous contrast also may help detect microadenomas, which then appear as focal hyperintense lesions relative to the surrounding gland.[8] This technique, however, is rarely used because delayed imaging is impractical.
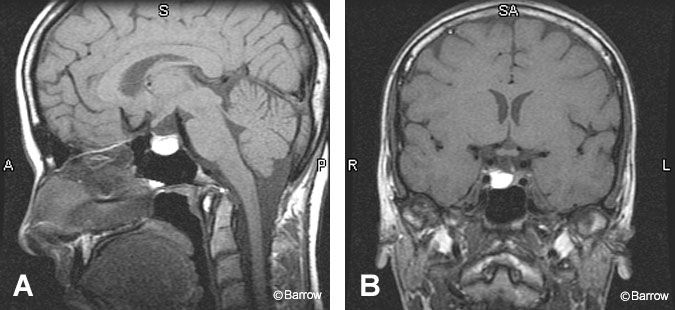
Pituitary Macroadenomas
Because macroadenomas are larger than 10 mm in diameter, they are easier to detect radiologically than microadenomas. These benign tumors often enlarge the sella turcica, a finding that can be appreciated on plain radiographs and cross-sectional imaging. On noncontrast T1-weighted imaging, these intrinsic pituitary tumors are hypointense to the surrounding gland. They exhibit heterogeneous enhancement after gadolinium administration. On T2-weighted sequences, they are more often hyperintense than microadenomas, particularly if they are soft or necrotic.[19] About 20 to 30% of adenomas show evidence of hemorrhage.[23] Subacute hemorrhage is best appreciated on T1-weighted sequences where it appears as hyperintense methemoglobin (Fig. 6). Signal intensity on T2-weighted imaging is variable. Typically, hemorrhage is asymptomatic although it may cause clinical symptoms of pituitary apoplexy.[18,21]
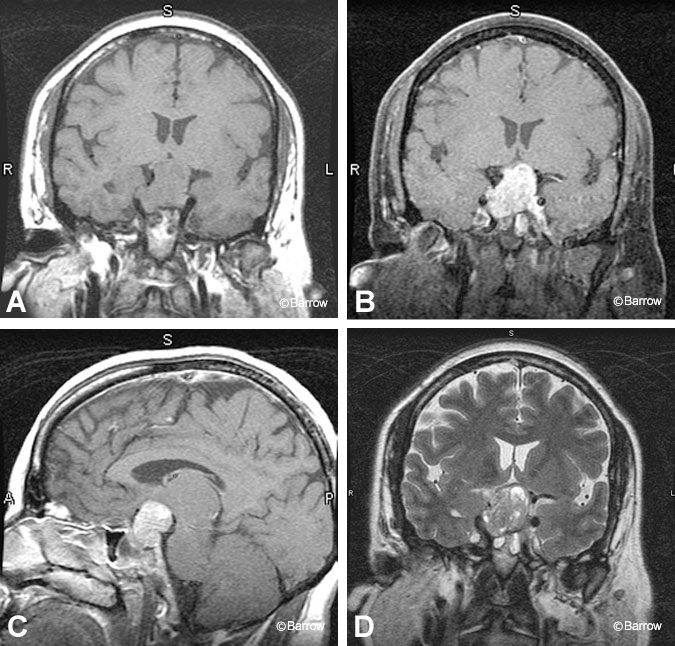
As the pituitary macroadenoma enlarges, it has a tendency to invade surrounding structures. Superior extension into the suprasellar cistern is well demonstrated on MR imaging, which allows compression of the optic chiasm to be assessed (Fig. 7). Tumor invasion of the cavernous sinus is more difficult to determine; traditionally, the presence of tumor lateral to the internal carotid artery is a reliable sign (Fig. 8).[6] Encasement of the intercavernous internal carotid artery by adenoma greater than or equal to 67% was concluded to be a specific sign of a cavernous sinus invasion in one study.[6] Extrinsic narrowing of the carotid artery is rarely associated with pituitary adenomas and is more suggestive of meningiomas. Tumor also may extend inferiorly into the sphenoid sinus or clivus with a predisposition to erode bone. As a result, the hyperintensity of the clival bone marrow on T1-weighted images is lost.
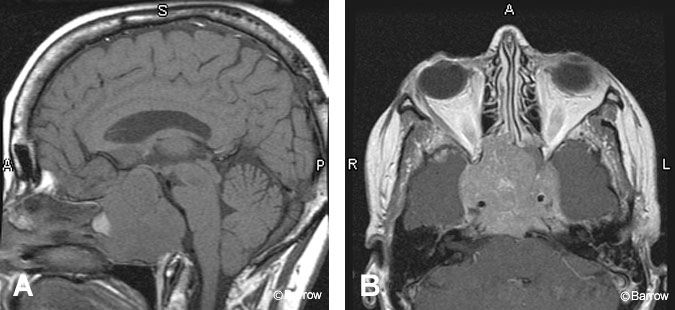
MR imaging is a useful technique for following macroadenomas. The appearance after transsphenoidal surgery depends on the packing materials used. Fat packed in the surgical defect appears hyperintense on T1-weighted sequences and requires the use of fat-saturated sequences to distinguish contrast enhancement from packing material. The MR imaging signal from Gelfoam is variable although it is often isointense to the pituitary gland with a hypointense center.[7] Both types of packing material show significant atrophy over time. Thus follow-up imaging is key for determining the presence of residual tumor.
Differential Diagnosis of Pituitary Tumors
There is a wide differential diagnosis for mass lesions within the sellar and suprasellar regions, including not only tumors but also inflammatory and vascular lesions.


Craniopharyngiomas and Rathke cleft cysts share the same embryological origin, the craniopharyngeal duct. Most craniopharyngiomas are centered on the suprasellar cistern. Occasionally, they are located entirely within the sella or third ventricle.[16] The classic form, the adamantinomatous craniopharyngioma, almost always contains cystic components, which appear hyperintense on T1-weighted images because of their high cholesterol content (Fig. 9). Adamantinomatous tumors also usually contain solid enhancing components as well as calcification, creating a heterogenous appearance on MR imaging. In contrast, papillary craniopharyngiomas, which are most often found in adults, are usually solid enhancing lesions in the third ventricle. Rathke cleft cysts are well-defined, round lesions often found in the pars intermedia of the pituitary gland. Their appearance on MR imaging reflects their composition. On T1-weighted images, they are hyperintense if they contain cholesterol-high mucoid material (Fig. 10) and hypointense if they contain serous fluid.[15] Calcification and enhancement are both extremely unusual.

About 10% of meningiomas occur in the parasellar region, arising from several locations including the tuberculum sellae, cavernous sinus, and sphenoid wing.[22] These tumors tend to lie somewhat eccentric to the sella, rather than arising from it. They are often associated with a thick adjacent dural tail. Although hyperintensity on T1- and T2-weighted images can be variable, meningiomas show strong homogeneous enhancement after contrast administration (Fig. 11). Associated hyperostosis and calcification can be detected by MR imaging but are better assessed by computed tomography (CT).
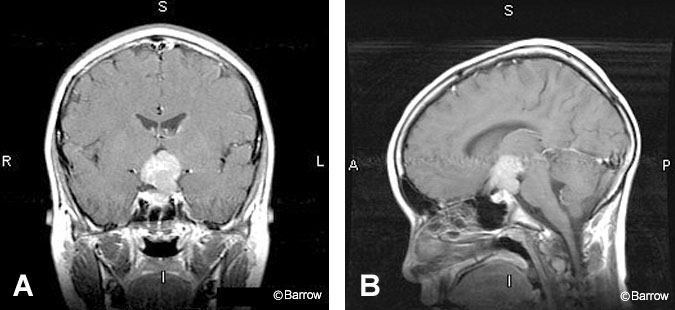
Germinomas are also strongly enhancing homogeneous tumors found in the suprasellar region (Fig. 12).[13] They may occur primarily in the suprasellar region or as a metastatic deposit from a pineal region tumor.[20] This midline germ-cell tumor can involve the infundibulum, hypothalamus, or anterior third ventricle and often appears hyperdense on noncontrast CT.
Epidermoid and dermoid tumors are benign tumors thought to arise from inclusion of epithelium during closure of the neural tube. Epidermoids are often located lateral to the midline and insinuate around adjacent structures. They are typically hyperintense to CSF on proton-density and diffusion-weighted imaging, which helps differentiate them from arachnoid cysts. Enhancement and calcification are both rare. Dermoid tumors are most often found in children and are located in the midline. They appear heterogeneous because of the presence of dermal appendages and squamous epithelium.

Metastatic disease can involve the pituitary gland, particularly in patients with advanced breast or bronchogenic carcinoma (Fig. 13). These lesions can occur in the sella or adjacent structures and are commonly associated with bone destruction.

Granular cell tumors are rare primary tumors located within the neurohypophysis. Although small nodules of granular cell proliferation are common in this region at autopsy, symptomatic tumors are extremely unusual. This tumor shows contrast enhancement on T1-weighted imaging and has a somewhat heterogeneous appearance on T2-weighted imaging (Fig. 14).
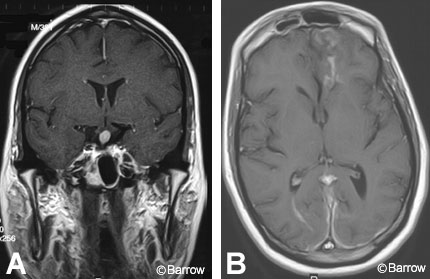
Inflammatory lesions such as lymphocytic hypophysitis and sarcoidosis cause thickening and enhancement of the infundibulum. Lymphocytic hypophysitis likely results from an autoimmune process and almost exclusively occurs in peripartum women. MR imaging shows diffuse enlargement of the anterior lobe of the pituitary gland associated with contrast enhancement.[12] On MR imaging, sarcoidosis tends to be associated with other intracranial findings, including multiple intraparenchymal or dural-based enhancing lesions and meningeal enhancement (Fig. 15).[5]
Aneurysms arising from the cavernous or supraclinoid segments of the carotid artery are extremely important lesions to distinguish from pituitary adenomas before surgery. On routine T1-weighted spin-echo sequences, aneurysms are hypointense due to the signal void produced by flowing blood.[2] When thrombosed, an aneurysm is hyperintense on T1-weighted images. T2-weighted images can help detect a laminated appearance, a useful diagnostic feature of aneurysms. If needed, MR angiography can be used to confirm the diagnosis.
Conclusion
MR imaging is the optimal imaging technique for assessing the sellar or suprasellar region. High-resolution imaging is required to evaluate the normal anatomy and to detect subtle tumors such as microadenomas. The differential diagnosis of pituitary adenomas is wide and includes not only other primary and secondary tumors but also inflammatory and vascular lesions.
References
- Ahmadi H, Larsson EM, Jinkins JR: Normal pituitary gland: Coronal MR imaging of infundibular tilt. Radiology 177:389-392, 1990
- Biondi A, Scialfa G, Scotti G: Intracranial aneurysms: MR imaging. Neuroradiology 30:214-218, 1988
- Chanson P, Daujat F, Young J, et al: Normal pituitary hypertrophy as a frequent cause of pituitary incidentaloma: A follow-up study. J Clin Endocrinol Metab 86:3009-3015, 2001
- Cheeman L, Kucharczyk W, Montanera WJ: Sella turcica and parasellar region, in Atlas SW (ed): Magnetic Resonance Imaging of the Brain. Philadelphia: Lippincott Williams and Wilkins, 2002, pp 1283-1362
- Cooper SD, Brady MB, Williams JP, et al: Neurosarcoidosis: Evaluation using computed tomography and magnetic resonance imaging. J Comput Tomogr 12:96-99, 1988
- Cottier JP, Destrieux C, Brunereau L, et al: Cavernous sinus invasion by pituitary adenoma: MR imaging. Radiology 215:463-469, 2000
- Dina TS, Feaster SH, Laws ER, Jr., et al: MR of the pituitary gland postsurgery: Serial MR studies following transsphenoidal resection. AJNR Am J Neuroradiol 14:763-769, 1993
- Dwyer AJ, Frank JA, Doppman JL, et al: Pituitary adenomas in patients with Cushing disease: Initial experience with Gd-DTPA-enhanced MR imaging. Radiology 163:421-426, 1987
- Elster AD, Chen MY, Williams DW 3rd, et al: Pituitary gland: MR imaging of physiologic hypertrophy in adolescence. Radiology 174:681-685, 1990
- Elster AD: Sellar susceptibility artifacts: Theory and implications. AJNR Am J Neuroradiol 14:129-136, 1993
- Fujisawa I, Asato R, Nishimura K, et al: Anterior and posterior lobes of the pituitary gland: Assessment by 1.5 T MR imaging. J Comput Assist Tomogr 11:214-220, 1987
- Hungerford GD, Biggs PJ, Levine JH, et al: Lymphoid adenohypophysitis with radiologic and clinical findings resembling a pituitary tumor. AJNR Am J Neuroradiol 3:444-446, 1982
- Jellinger K: Primary intracranial germ cell tumours. Acta Neuropathol (Berl) 25:291-306, 1973
- Kucharczyk W, Davis DO, Kelly WM, et al: Pituitary adenomas: High-resolution MR imaging at 1.5 T. Radiology 161:761-765, 1986
- Kucharczyk W, Peck WW, Kelly WM, et al: Rathke cleft cysts: CT, MR imaging, and pathologic features. Radiology 165:491-495, 1987
- Matthews FD: Intraventricular craniopharyngioma. AJNR Am J Neuroradiol 4:984-985, 1983
- Nakamura T, Schorner W, Bittner RC, et al: The value of paramagnetic contrast agent gadolinium-DTPA in the diagnosis of pituitary adenomas. Neuroradiology 30:481-486, 1988
- Ostrov SG, Quencer RM, Hoffman JC, et al: Hemorrhage within pituitary adenomas: How often associated with pituitary apoplexy syndrome? AJR Am J Roentgenol 153:153-160, 1989
- Snow RB, Johnson CE, Morgello S, et al: Is magnetic resonance imaging useful in guiding the operative approach to large pituitary tumors? Neurosurgery 26:801-803, 1990
- Takeuchi J, Handa H, Nagata I: Suprasellar germinoma. J Neurosurg 49:41-48, 1978
- White W, Liu SS: Pituitary apoplexy, in Carter LP, Spetzler RF, (eds): Neurovascular Surgery. New York: McGraw-Hill, 1995
- Yeakley JW, Kulkarni MV, McArdle CB, et al: High-resolution MR imaging of juxtasellar meningiomas with CT and angiographic correlation. AJNR Am J Neuroradiol 9:279-285, 1988
- Yousem DM, Arrington JA, Zinreich SJ, et al: Pituitary adenomas: Possible role of bromocriptine in intratumoral hemorrhage. Radiology 170:239-243, 1989
