
Early Cognitive and Affective Sequelae of Traumatic Brain Injury: A Study Using the BNI Screen for Higher Cerebral Functions*
Authors
Susan R. Borgaro, PhD
George P. Prigatano, PhD
Division of Neurology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
*Adapted from Borgaro SR, Prigatano GP: Early Cognitive and Affective Sequelae of Traumatic Brain Injury: A Study Using the BNI Screen for Higher Cerebral Functions. J Head Trauma Rehabilitation 17:526-534, 2002. Adapted with permission from Lippincott Williams & Wilkins.
Abstract
The BNI Screen for Higher Cerebral Functions (BNIS) was used to assess how cognitive and affective disturbances can be differentially influenced during the acute stages of traumatic brain injury (TBI). Forty-two patients with a moderateto-severe TBI were administered the BNIS within the first 60 days of injury. Their performance was compared to a convenience sample of 21 control subjects used in the standardization of the BNIS. Compared to patients with TBI, control subjects performed significantly better on the BNIS total score and on all subtest scores. TBI patients were best classified by poor performance on measures of affect disturbance and impaired awareness. Stepwise discriminant analysis identified that disturbances in memory, awareness, and affect contributed most to the classification of an individual as having a TBI. During the early stages after significant TBI, the BNIS can be used to assess both cognitive and affective disturbances directly. This brief screening tool also can help document that the early stages of TBI specifically affect memory, awareness, and affect.
Key Words: acute rehabilitation, cognitive and affective disturbances, traumatic brain injury
The nature of higher cerebral functions is not purely “cognitive.” They often reflect an integration of thinking and feeling.[19] Moderate-tosevere traumatic brain injury (TBI) thus produces disturbances in both realms. In their early work, Chapman and Wolff[4] noted that brain dysfunctional patients show not only problems with memory, judgment, and abstract reasoning but also a reduced tolerance for frustration, inappropriate affective reactions, lack of spontaneity, and diminished seeking of nonadaptive states (e.g., challenges, adventure, or exploration).
Typically, neuropsychologists have focused on cognitive deficits after TBI.[6,7,11,13,31,35,36] Speed of information processing and memory deficits are commonly studied[5,7,12] along with language disturbances.[31] Affective disturbances are often noticed but seldom assessed directly. A recent collaborative project, for example, used traditional methods of assessing motor speed, language disturbance, visuospatial abilities, abstract reasoning, and memory when attempting to relate early cognitive deficits to later outcome after TBI. Emotional and motivational disturbances were not studied.[3]
Typically, when affective disturbances are assessed, questionnaires or rating scales are used.[14-16] There are, however, a few exceptions. For example, St. Clair and colleagues[34] tested patients with Parkinson’s disease on facial and word emotional identification tasks and on traditional tests of memory, visual attention, and naming. Unfortunately, the clinical application of this type of information is often lacking, and it is rarely used specifically to justify or plan rehabilitation interventions.
Postacute holistic rehabilitation programs for TBI patients have documented the importance of affective disturbances in determining rehabilitation outcomes.[2,9,23,29] Recently, Prigatano and Wong[26] related cognitive and affective impairments to the achievement of rehabilitation goals during the early stages after TBI and cerebrovascular accidents. In these days of managed health care, it is important to document both affective and cognitive disturbances to help justify prescribed rehabilitation services. Doing so in a cost-effective manner is becoming progressively important.[24]
The BNI Screen for Higher Cerebral Functions (BNIS) was designed to assess both cognitive and affective disturbances briefly and validly, particularly for patients in the early stages of brain injury. Construct validity of the test in postacute TBI patients is good.[37] In addition to assessing five cognitive domains (speech and language, orientation, attention/concentration, visuospatial and visual problem solving, and learning and memory) and awareness (awareness vs. performance), the BNIS attempts to measure affective responses directly. Patients are asked to read a sentence with both a happy and angry tone of voice (affect expression) and to identify verbally three facial expressions (angry, happy, and fear-surprise). They are also presented stimuli designed to provoke spontaneous laughter or amusement.
The present investigation extends our previous observations[26] by focusing on how severity of TBI influences both cognitive and affective functions as well as awareness[17] during the early stages after a moderate-to-severe TBI. We predicted that patients with a moderate or severe TBI would perform below average on tests of both cognitive and affective functioning. We explore how cognitive, affective, and awareness disturbances may be differentially affected by moderate-to-severe TBI during the early stages following such injuries. The overall goals were to demonstrate that a brief neuropsychological assessment can sample both dimensions and that this information is potentially useful when documenting the needs of such patients during the early stages of brain injury rehabilitation.
Method
Subjects
Forty-two patients with an admitting diagnosis of cerebral concussion or contusion (based on codes from the International Classification of Diseases) were the experimental subjects (Table 1). Based on their diagnostic imaging studies and admitting Glasgow Coma Scale (GCS) scores,[10] which were obtained in the emergency room, the 42 patients were classified as having a severe or moderate brain injury. All participants with a TBI were referred for a clinical neuropsychological evaluation as a part of their neurological and/or rehabilitation evaluation.
The inclusion criteria for TBI patients were as follows: (1) Participants were within 60 days of their documented TBI. (2) Their admitting GCS score and neuroimaging studies qualified the severity of their injury. (3) Participants were able to take a brief screening test as part of their clinical evaluation. (4) Participants had no history of either a prior TBI or significant psychiatric illness. (5) Finally, participants spoke English as their primary language. All subjects were interviewed and administered the BNIS by either a staff clinical neuropsychologist or a postdoctoral resident in clinical neuropsychology.
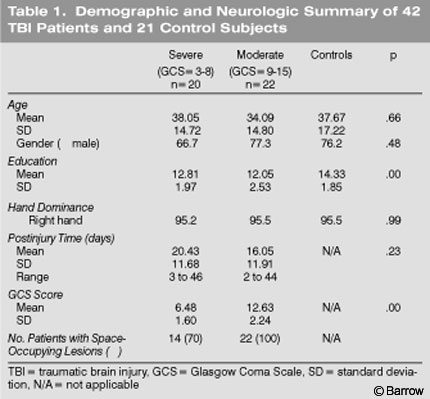
Twenty patients with severe TBIs (Table 1) had admitting GCS scores between 3 and 8. Fourteen (70%) had space-occupying lesions as reflected by computed tomography (CT) and magnetic resonance (MR) imaging (which were recorded but not deemed necessary to classify a TBI as severe). Of these 14 patients, 19% had bilateral lesions, 31% had left hemisphere lesions, and 31% had right hemisphere lesions. Another 19% had a space-occupying lesion in the brain stem, cerebellum, or an unspecified location. The most common lesions were contusions (65%),hemorrhages (26%),and hematomas (9%).
Twenty-two patients had moderate TBIs (Table 1) as defined by an admitting GCS score between 9 and 12 or by a GCS score between 13 and 15 and the presence of a space-occupying lesion. At admission, all 22 patients had a space-occupying lesion on CT or MR imaging (Table 1). Of the 22 patients, 36% had bilateral lesions, 27% had left hemisphere lesions, and 23% had right hemisphere lesions. In 14% of the patients, the lesion was in either the brain stem, cerebellum, or an unspecified location. The most common lesions were contusions (58%), hemorrhages (28%), and hematomas (14%,Table 1).
Twenty-one control subjects, initially recruited as a part of the standardization study of the BNIS, also were administered the BNIS (Table 1). Control subjects reported no history of psychiatric or neurologic disease.
Procedures
Patients were evaluated within the first 60 days of their injury. All subjects were interviewed and administered the BNIS either by a staff clinical neuropsychologist or by a postdoctoral resident in clinical neuropsychology.
Neuropsychological Instrument
The BNIS was designed to provide a brief but valid assessment of higher cerebral functions from acute to chronic stages of various brain disorders. Initial studies have demonstrated its inter-rater and test-retest reliability[27] as well as its concurrent and construct validity.[20,28] This test and its various subtests reliably differentiate brain dysfunctional patients from patients with psychiatric disorders and from patients with acute medical illness.[28]
The BNIS typically requires 10 to 25 minutes to administer. Scores obtained from the BNIS include a total score (range of raw scores = 3 to 50 points) and seven subtest scores. Maximum subtest scores range from 1 to 15, depending on the number of items that compose the scale. Higher scores indicate a higher level of functioning. The following briefly describes each of the seven subtests (for a detailed description of the individual items, see Prigatano et al.[21] and Rosenstein et al.[28]): (1) the Speech and Language subtest consists of 13 items that assess primary and secondary language abilities;(2) the Orientation subtest consists of three items that assess orientation to time and place and right-left orientation; (3) the Attention/Concentration subtest consists of three items that assess the ability to attend to simple and complex auditory information; (4) the Visuospatial and Visual Problem Solving subtest consists of seven items that assess the ability to scan, sequence, and process visual information; (5) the Learning and Memory subtest consists of learning and remembering four-number symbol associates as well as three words with distraction; (6) the Affect subtest consists of four items designed to assess the ability to express happy and angry tones of voice verbally when reading a sentence, to perceive facial affect, to control affect during testing, and to demonstrate spontaneous affect; (7) the Awareness vs. Performance subtest consists of a single item that assesses the difference between predicted and actual performance on a task of memory recall.
In addition to the seven subtest items, three prescreening items (level of consciousness/alertness, basic language, level of cooperation) that contribute to the BNIS total score but not to any of the subtest scores were administered. The BNIS is not administered if the patient does not demonstrate adequate alertness, language ability, or cooperation on the prescreening items. The BNIS total (raw) score is converted to age-corrected standard (T) scores using the norms of Prigatano and colleagues.[21] Subtests scores are not converted to T scores.
Data Analysis
Demographic data were examined for group differences. One-way analyses of variance (ANOVAs) were conducted to determine differences in age, education, and time since injury among the groups. Gender differences among the groups were analyzed by Chi-square analysis. Differences among the three groups in terms of scores on the BNIS subtests scores were then analyzed by ANOVA. Multiple comparisons of groups were controlled by using the Bonferroni correction with an alpha level of p<0.007 considered statistically significant. Analyses of covariance (ANCOVA) were performed when groups differed on variables shown to influence performance on neuropsychological tests. Finally, a stepwise discriminant function analysis was computed to determine if any of the BNIS subtests could differentiate the groups from each other.
Results
Demographic Comparisons
The TBI and control groups did not differ in terms of age [F(2,61)= 0.42, p= 0.657] or gender (x[2]= 0.74, p=0.690). The control group, however, had a significantly higher level of education than the two TBI groups [F(2,61)=6.29, p= 0.003, Table 1]. The BNIS total score correlated significantly with years of education [r= 0.47, p<0.001].[21] This variable was therefore used as a covariate to analyze group differences on the BNIS. Time of testing after injury did not differ between the two TBI groups [F(1,40)= 1.45,p=0.236].
BNI Scores and TBI Severity
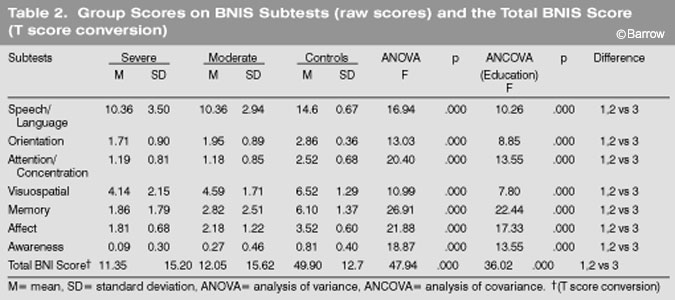
Both TBI groups performed significantly worse (p<0.001) than the control group on the BNI Total Score (T score value) and on all seven of the BNIS subtests (Table 2). When the analyses were recalculated using education as a covariate, the differences were again significant (p<0.001).
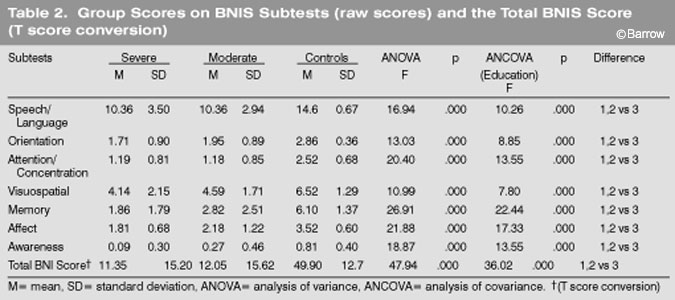
A trend was observed across BNI subtest scores and group severity. Patients with severe TBIs performed worse than patients with moderate TBIs who performed worse than controls on all measures of the BNIS. However, the mean differences between the severe and moderate groups during the first 46 days after injury were not statistically significant (Table 2).
Cognitive and Affective Variables as Predictors of TBI
Because the performance of the two TBI groups did not differ on any of the BNIS subtests, a stepwise discriminant function analysis was performed to determine if any of the cognitive or affective subtests could significantly differentiate the TBI groups from the control group.
The two TBI groups were pooled and compared to the control group on all of the BNIS subtests. One discriminant function was calculated (x[2]= 51.78, p<0.001) and accounted for 100% of the total variance. The only subtests that loaded on this factor were memory, awareness, and affect, respectively (Table 3).
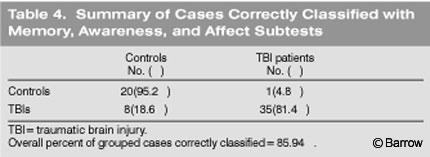
Overall, the discriminant function based on Wilks Lambda correctly classified controls and patients on these three of the seven subtests 86% of the time. These items correctly classified the TBI group 81.4% of the time and the control group 95.2% of the time (Table 4).
Discussion
This study documented the wide range of neuropsychological disturbances observed in patients with a moderate-to-severe TBI during the acute phases of their injury. On measures of both cognitive and affective functions, patients with a TBI performed worse than controls. This difference remained reliable after differences in educational level between the controls and TBI subjects were statistically covaried. Although a “dose-response relationship” trend was observed between severity of injury and neuropsychological test performance, group differences between moderate and severely injured patients were not statistically significant. This finding may partially reflect the acuteness and severity of the patients’ injuries. Not surprisingly, patients with both moderate and severe TBIs scored considerably below average compared to the control group. The test performance of both groups of patients during the early stages of injury may reflect considerable bilateral cerebral dysfunction despite the differences in their GCS admitting scores and would account for their low level of performance. Assessing these patients 6 to 12 months after injury might reveal a clearer dose-response relationship. When such relationships have been reported, they have tended to involve patients 1 year after injury.[7]
When the TBI groups were combined and compared to the control group, disturbances in memory, awareness, and affect most differentiated the two groups. Numerous studies have documented the variety of cognitive deficits associated with severe and moderate TBI, and memory disturbance has been among the most common. [5,7] The present study reaffirms this finding and also documents the importance of affective disturbances. Although changes in affect, mood, and personality have long been recognized after various forms of brain injury,[4,5,18,37] few clinical studies have attempted to measure these changes directly with neuropsychological tests.
In addition to demonstrating problems in memory and affect, the present sample of TBI patients also showed impairment on a single measure used to elicit disturbances of awareness. Although disturbances of awareness are common after various forms of brain injury[10,18,19,25] and have been noted specifically during the rehabilitation of postacute TBI patients,[19,23] they have not been easily associated with other measures of cognitive impairment. Sherer et al.[32] and Prigatano et al.,[22] however, have documented that clinicians’ judgments of patients’ impaired awareness are related to the severity of TBI as measured by GCS scores. Sherer et al.[33] have shown that early impaired awareness after TBI is, in fact, associated with employability after rehabilitation. Collectively, these findings suggest that impaired awareness is an important area to assess during every stage following TBI.
Implications for Early Cognitive Remediation of TBI Patients
The present observations suggest that early cognitive rehabilitation for TBI patients should focus not only on their cognitive deficits but on their affective disturbances as well. The findings suggest that working with patients to improve the accuracy of their predictions (perhaps teaching them improved self-awareness) and helping them to perceive affect, to generate affect, and to show spontaneous affect may be important in early cognitive interventions. Focusing on the specific cognitive and affective disturbances of TBI patients may produce better outcomes than those associated with using a standard protocol of cognitive rehabilitation for all patients.[30] The findings also reaffirm the importance of working with this population on such traditional areas of dysfunction as memory impairment.
Limitations of Study
The difference in educational level between controls and TBI patients is a limitation of this study. Although this variable was partially controlled through the analysis of covariance and our findings remained highly robust, this statistical procedure has limitations. In addition, studies with normal elderly[8] and patients with dementia of the Alzheimer’s type1 have suggested that education plays a protective role in the emergence of neuropsychological symptoms. Whether education also exerts a protective influence in the recovery after severe and moderate TBI needs to be assessed further.
Although there were no significant differences between the two TBI groups in terms of time since injury, patients with severe injuries were examined a mean of 20 days after injury while patients with moderate injuries were examined 16 days after injury. Severity of injury could interact with time since injury to produce different neuropsychological test findings.[21] How educational level, acuteness of injury, and severity of injury interact to produce this symptom picture also needs to be explored further.
Conclusions
This study argues that neuropsychological evaluations should directly assess both cognitive and affective disturbances after TBI. Patients can be assessed early after injury using measures such as the BNIS. Such assessments may assist in patient management and help to document disturbances in cognition, affect, and awareness. Providing such information can be used to improve patient management and to predict rehabilitation outcomes.[19,26,33]
References
- Alexander G, Furey M, Grady C, et al: Association of premorbid intellectual function with cerebral metabolism in Alzheimer’s disease: Implications for the cognitive reserve hypothesis. Am J Psychiatry 154:165-172, 1997
- Ben-Yishay Y, Prigatano G: Cognitive remediation, in Rosenthal M, Bond M (eds): Rehabilitation of the Adult and Child with Traumatic Brain Injury. Philadelphia: F.A. Davis, 1990, pp 393-409
- Boake C, Millis SR, High WM, Jr., et al: Using early neuropsychologic testing to predict longterm productivity outcome from traumatic brain injury. Arch Phys Med Rehabil 82:761-768, 2001
- Chapman L, Wolff H: The cerebral hemispheres and the highest integrative functions of man. AMA Arch Neurol 1:357-424, 1959
- Ciba Foundation Symposium 34: Outcome of Severe Damage to the Central Nervous System. Amsterdam: Elsevier Scientific, 1975
- Crosson B, Sartor K, Jenny A, et al: Increased intrusions during verbal recall in traumatic and nontraumatic lesions of the temporal lobe. Neuropsychology 7:193-208, 1993
- Dikmen SS, Manchamer J, Winn H, et al: Neuropsychological outcome at 1-year post head injury. Neuropsychology 9:80-90, 1995
- Evans D, Beckett L, Albert M, et al: Level of education and change in cognitive function in a community population of older persons. Ann Epidemiol 3:71-77, 1993
- Ezrachi O, Ben-Yishay Y, Kay T, et al: Predicting employment in traumatic brain injury following neuropsychological rehabilitation. J Head Trauma Rehabil 6:71-84, 1991
- Jennett B, Teasdale G: Management of Head Injuries. Philadelphia: F.A. Davis, 1974
- Johnson S, Bigler E, Burr R, et al: White matter atrophy, ventricular dilation, and intellectual functioning following traumatic brain injury. Neuropsychology 8:307-315, 1994
- Levin H, Gary HE, Jr., Eisenberg H, et al: Neurobehavioral outcome 1 year after severe head injury. Experience of the Traumatic Coma Data Bank. J Neurosurg 73:699-709, 1990
- Levin H, Mendelsohn D, Lilly M, et al: Tower of London performance in relation to magnetic resonance imaging following closed head injury in children. Neuropsychology 8:171-179, 1994
- Lezak M: Neuropsychological Assessment. New York: Oxford University, 1995
- Mathias J, Coats J: Emotional and cognitive sequelae to mild traumatic brain injury. J Clin Exp Neuropsychol 21:200-215, 1999
- McCleary C, Satz P, Forney D, et al: Depression after traumatic brain injury as a function of Glasgow Outcome Score. J Clin Exp Neuropsychol 20:270-279, 1998
- Prigatano G: Disturbances of self-awareness of deficit after traumatic brain injury, in Rosenstein L, Prigatano G, Amin K (eds): Awareness of Deficit After Brain Injury. New York: Oxford University, 1991, pp 111-126
- Prigatano G: Personality disturbances associated with traumatic brain injury. J Consult Clin Psychol 60:360-368, 1992
- Prigatano G: Principles of Neuropsychological Rehabilitation. New York: Oxford University, 1999
- Prigatano G, Amin K, Rosenstein L: Validity studies on the BNI Screen for Higher Cerebral Functions. BNI Quarterly 9(1):2-9, 1993
- Prigatano G, Amin K, Rosenstein L: Administration and Scoring Manual for the BNI Screen for Higher Cerebral Functions. Phoenix, AZ: Barrow Neurological Institute, 1995
- Prigatano G, Bruna O, Mataro M, et al: Initial disturbances of consciousness and resultant impaired awareness in Spanish patients with traumatic brain injury. J Head Trauma Rehabil 29-38, 1998
- Prigatano G, Fordyce D, Zeiner H, et al: Neuropsychological Rehabilitation After Brain Injury. Baltimore: John Hopkins University, 1986
- Prigatano G, Pliskin N: Clinical Neuropsychology and Cost Outcomes Research: A Beginning. New York: Psychology Press, 2002
- Prigatano G, Schacter D: Awareness of Deficit After Brain Injury: Clinical and Theoretical Issues. New York: Oxford University, 1991
- Prigatano G, Wong J: Cognitive and affective improvements in brain dysfunctional patients who achieve inpatient rehabilitation goals. Arch Phys Med Rehabil 80:77-84, 1999
- Rosenstein L, Prigatano G, Amin K: Reliability studies for the BNI Screen for Higher Cerebral Functions. BNI Quarterly 8(3):24-28, 1992
- Rosenstein L, Prigatano G, Nayak M: Differentiating patients with higher cerebral dysfunction from patients with psychiatric or acute medical illness using the BNI Screen for Higher Cerebral Functions. Neuropsychiatry Neuropsychol Behav Neurol 10:113-119, 1997
- Rottok J, Ben-Yishay Y, Lakin P, et al: Outcome of different treatment mixes in a multidimensional neuropsychological rehabilitation program. Neuropsychology 6:395-415, 1992
- Salazar A, Warden D, Schwab K, et al: Cognitive rehabilitation for traumatic brain injury: A randomized trial. Defense and Veterans Head Injury Program (DVHIP) Study Group. JAMA 283:3075-3081, 2000
- Sarno M, Buonaguro A, Levita E: Characteristics of verbal impairment in closed head injured patients. Arch Phys Med Rehabil 67:400-405, 1986
- Sherer M, Boake C, Levin E, et al: Characteristics of impaired awareness after traumatic brain injury. J Int Neuropsychol Soc 4:380-387, 1998
- Sherer M, Hart T, Nick TG, et al: Early impaired self-awareness after traumatic brain injury. Arch Phys Med Rehabil 84:168-176, 2003
- St.Clair J, Borod J, Sliwinski M, et al: Cognitive and affective functioning in Parkinson’s disease patients with lateralized motor signs. J Clin Exp Neuropsychol 20:320-327, 1998
- van Zomeren A, van den Berg W: Residual complaints of patients two years after severe head injury. J Neurol Neurosurg Psychiatry 48:21-28, 1985
- Vilkki J: Cognitive flexibility and mental programming after closed head injuries anterior or posterior cerebral excisions. Neuropsychologia 30:807-814, 1992
- Waas P: An Analysis of the Construct-Related and Ecological Validity of the Barrow Neurological Institute Screen for Higher Cerebral Functions (doctoral dissertation). Ontario, Canada: University of Windsor, 1997
