
Brain Activation Patterns in the Affected and Nonaffected Hand After Subcortical Stroke: Two Case Reports Using Serial fMR Imaging
George P. Prigatano, PhD
Shawn Gale, PhD
Leslie Baxter, PhD
Sterling Johnson, PhD*
Division of Neurology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona *Current address: William S. Middleton VA Hospital, Madison, Wisconsin
Abstract
Serial fMR imaging correlates of finger tapping were obtained in two middle-aged men after subcortical strokes that affected motor performance with their left hand. Brain activation patterns were obtained when both the “affected” and the “nonaffected” hand performed a modified version of the Halstead Finger Tapping Test. Improved speed of finger tapping in the affected hand (Case 1) was associated with widespread bilateral cerebral activation and ipsilateral cerebellar activation. This pattern did not appear in the second patient who was unable to tap with his affected hand. Bilateral cerebral and ipsilateral cerebellar activation was observed when both patients tapped with their unaffected hand, but to a lesser extent in the patient with poor motor recovery in the affected hand. The findings emphasize two points. First, there is a dynamic interaction among the two cerebral hemispheres and cerebellum during the recovery process. Second, the cerebellum may play an important role in the initiation of finger movement. Failure to show normal patterns of brain activation after stroke while performing a motor task may be a marker of “less efficient” brain functioning. It is associated with a subjective sense of fatigue, which is often reported by patients who have had a stroke.
Key Words: cerebrovascular accident, finger tapping, functional magnetic resonance imaging, recovery, stroke
Abbreviations Used: CVA, cerebrovascular accident; fMR, functional magnetic resonance; MPSPGR, multiplanar spoiled gradient; NEX, number of excitations; TE, echo time; TR, pulse repetition time
Moore[11] has emphasized that we tend to forget “the bilateral nature of the nervous system” and that “everything man does on one side of the body immediately affects and is affected in turn by the opposite side.” Behavioral studies using the Halstead Finger Tapping (or Oscillation) Test to study individuals with unilateral cerebral lesions support this observation. For example, after a unilateral CVA that affects motor functioning, speed of finger tapping is often affected in the hand contralateral to the lesion.[14] Speed of finger tapping also can be mildly slowed in the hand ipsilateral to the lesion.[12] Furthermore, a return to normal or near-normal speeds of finger tapping with both the affected and the so-called “unaffected” hand has been associated with achieving rehabilitation goals during the early stages after a CVA (i.e., within the first 30 days).[12] Mechanisms responsible for motor recovery after stroke remain unclear.[3] If understood, the information might help guide early and later efforts at motor rehabilitation.Johnson and Prigatano[7] had eight normal, healthy adults perform a version of the Halstead Finger Tapping Test while undergoing whole-brain echo-planar imaging. The motor map obtained was typical of reports of simple hand-motor experiments.[13] Activation patterns were greatest in the contralateral motor cortex and ipsilateral cerebellum. Activation was also observed in the supplementary motor area and ipsilateral motor cortex, but to a lesser extent.
Is this pattern affected after a CVA that produces hemiparesis or hemiplegia? Does this pattern change when motor recovery is observed compared to when it is not? Following Moore’s[11] line of reasoning, different patterns of cerebral activation would be expected in individuals who have made a good or poor behavioral recovery when performing the Halstead Finger Tapping Test.
To explore this hypothesis, we followed Cramer’s and Bastings’ suggestion that “serial studies within the same patient may have particular value” in understanding “the timing and site of many of the events relevant to clinical improvement after stroke.”[3] We therefore used fMR imaging to study two middle-aged men who suffered motor disturbances of the left hand after CVAs. The first patient made an excellent behavioral recovery. His speed of finger tapping returned to normal in the unaffected hand and partially recovered in the affected hand. Speed of finger tapping in the second patient returned to normal in the unaffected hand, but he was unable to produce even a single tap on the Halstead Finger Tapping Test with his affected hand. fMR imaging techniques were used to observe patterns of activation when the patients performed a version of the Halstead Finger Tapping Test on two separate occasions. Patterns of activation in both subjects were also studied on a similar, but different finger tapping test 3 years later to determine the consistency (or reliability) of the earlier observations.
Two questions were asked: (1) Is a different pattern of brain activation associated with partial recovery of function in the affected hand, as measured by finger- tapping, compared to no recovery? (2) Do different patterns of brain activation occur when the “unaffected” hand performs the Halstead Finger Tapping Test with different levels of motor recovery in the affected hand?
Materials and Methods
Patient 1
A 60-year-old, right-handed man suffered a right pontine infarction, which was followed by left hemiparesis, dysarthria, and decreased balance. He had 18 years of formal education with no history of a learning disability. He was studied using fMR imaging procedures about 15 days after his CVA (i.e., Time 1) and again 6 months later (Time 2). A third fMR imaging study using a 3 Tesla magnet and an alternative form of the Halstead Finger Tapping Test (discussed below) was obtained about 35 months later (i.e., approximately 3.5 years after the onset of his CVA).
Patient 2
A 52-year-old, right-handed man suffered an infarction in the right posterior limb of the internal capsule and subsequently developed left hemiparesis, left facial droop, and associated left hemiparesthesias. He had 12 years of formal education with no history of learning disability. He was first studied 9 days after his CVA (Time 1) and again 3 months later (Time 2). About 39 months after his CVA, a third fMR imaging study, using the 3 Tesla magnet and an alternative form of the Halstead Finger Tapping Test, was obtained.
Image Acquisition
During the first two fMR imaging studies, both participants underwent scanning on the same 1.5 Tesla, GE NVMR scanner. fMR imaging data were obtained using a gradient-echo, echo-planar pulse sequence with the following parameters: TE=40 ms, TR=3000 ms, flip angle=90 degrees, matrix=64 x 64 cm, field of view =240 mm, slice thickness=3.8 mm, and repetitions=92, yielding 36 contiguous axial slices of the brain at a near isotropic voxel resolution of 3.75 x 3.75 x 3.8 mm. A whole-brain T1-weighted SPGR structural image was obtained with the following parameters: TR=24, TE=6, flip angle=40 degrees, NEX=1, slice thickness=1.5 mm, 0 skip between slices, field of view=240 mm, and in-plane resolution=.9375 mm2 voxels.
The third scan was performed on a 3 Tesla GE scanner. Images were obtained using a gradient-echo, echo-planar pulse with the following parameters: TE=30 ms, TR=3000 ms, flip angle=90 degrees, matrix=64 x 64 and slice thickness=4 mm, yielding 32 contiguous axial slices at a voxel resolution of 3.75 x 3.75 x 4 mm. Whole-brain T1-weighted MPSPGR images were obtained with the following parameters: TE=min/full, prep time=300 ms, flip angle=30 degrees, NEX=2, slice thickness=2, 0 skip between slices, field of view=240 mm, and in-plane resolution=.9375 mm2 voxels.
fMR Imaging Task
Auditory and visual stimuli were presented using MRI-compatible goggles and headphones (Resonance Technology, Northridge, CA). A modified finger tapping procedure was used. During the first two scanning procedures, subjects tapped to a 1 Hz tone with their index finger. Each trial was 15 seconds and alternated with 15 seconds of rest. During each scanning session, five cycles (15 seconds tapping, 15 seconds resting) were completed with the right hand followed by five cycles with the left hand.
For the third scanning session performed with the 3 Tesla scanner, subjects tapped to a 1 Hz tone for pseudorandom blocks of right, left, and rest conditions. Each block was 18 seconds long, and 5 cycles per condition were performed during a session. Individual analyses comparing tapping with the right and left hand were generated using statistical parametric mapping (SPM99).
Neuropsychological Test Scores
Soon after their CVA, both subjects were administered neuropsychological tests (i.e., subtests of the Wechsler Adult Intelligence Scale, 3rd edition, Trail Making Test–Parts A and B, Controlled Oral Word Association Subtest of Multi-lingual Aphasia Test). When the patients returned for their third fMR imaging study, neuropsychological tests were again administered to determine their level of higher cerebral functioning. Patient 1 was tested as a part of his clinical follow-up. Patient 2 was tested only for the purpose of research. On each of the three occasions, the finger-tapping scores were obtained outside the fMR imaging scanner.
Results
Behavioral and Psychometric Observations
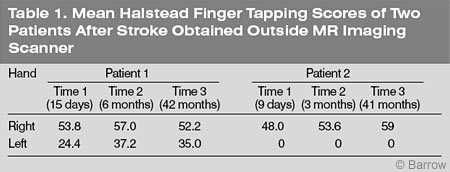
Using his right (unaffected) hand, Patient 1 finger tapped at a normal speed on the three occasions (Table 1). Tapping with his left (affected) hand was moderately affected within the first 2 weeks of his CVA but progressively improved over the next 5 weeks. Six months after his CVA, his speed of finger tapping with his left hand was within 1.5 standard deviations of normal, according to the norms provided by Heaton, Grant, and Matthews.[5]
In Patient 2, speed of finger tapping with his right (unaffected) hand appeared to be mildly affected after his CVA (Table 1). Three months after the onset of his CVA, however, speed of finger tapping with his right hand had returned to a normal range. This pattern of performance was maintained 3.5 years later. However, Patient 2 was unable to finger tap with his left (affected) hand on any of the three occasions.
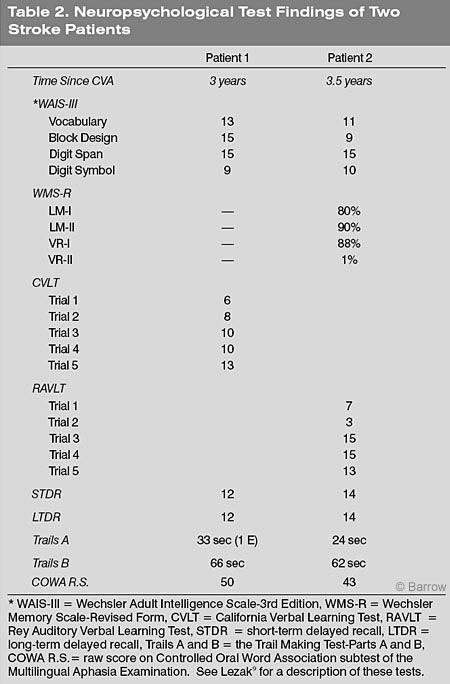
About 3.5 years after their CVA, both subjects had normal intellectual test scores, as measured by selected subtests of the Wechsler Adult Intelligence Scale-3rd Edition (Table 2). Patient 1 was not given portions of the Wechsler Memory Scale-Revised Form at this time. However, Patient 2’s performance on the Memory Scale was compatible with his known lesion. He had notable difficulty in the delayed recall (after 20 minutes) of visuospatial information. His performance was otherwise within the normal range. Using different tests of rote verbal learning, both subjects’ performance on rote verbal learning and delayed recall was normal. By the third fMR imaging session, both subjects’ performances were also normal on the Trail Making Test-Parts A and B and on the Controlled Oral Word Association subtest of the Multilingual Aphasia Test.
fMR Imaging Observations
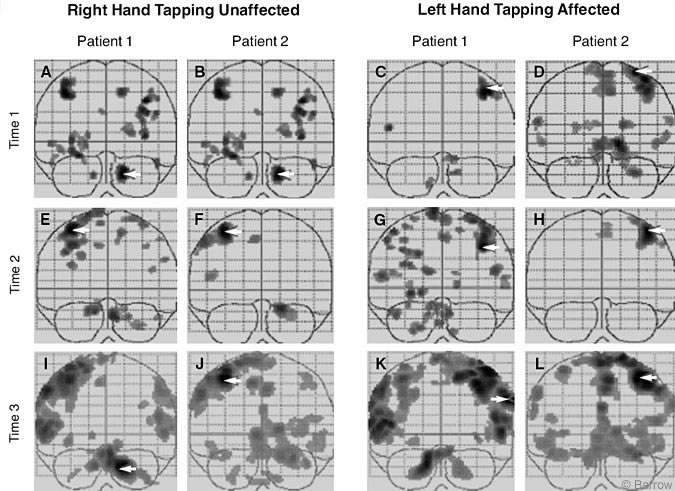
1. Are there different patterns of brain activation when partial motor recovery of the affected hand occurs, as measured by speed of finger tapping? Soon after the onset of his stroke, Patient 1 showed minimal activation while finger tapping with his left (affected) hand (Figs. 1C and 2C). At Time 2 (Figs. 1G and 2G), after some behavioral recovery, finger tapping with his left hand was associated with greater bilateral activation throughout the cerebral cortex than at Time 1. There was evidence of cerebellar activation at both times. The same pattern was maintained at Time 3, 3.5 years after the onset of his stroke. The increase in regional activation likely reflected the stronger signals obtained with the 3 Tesla scanner (Figs. 1K and 2K).
The opposite pattern was observed for Patient 2 whose affected (left) hand failed to recover. Initially, Patient 2 showed bilateral activation of the cerebral cortex and some activation in the cerebellum (Figs. 1D and 2D). When Patient 2 attempted to finger tap at Time 2, however, both hemispheres showed less activation (Figs. 1H and 2H) than at Time 1. The contralateral sensorimotor strip was activated, but almost no activation of the cerebellum was seen. When Patient 2 was tested using the 3 Tesla magnet 3.5 years after lesion onset (Figs. 1L and 2L), the same activation pattern was obtained.
2. Are there different patterns of brain activation when the unaffected hand performs a finger tapping test, but the affected hand has not recovered? Patient 1 showed clear evidence of bilateral activation when finger tapping immediately (Figs. 1A and 2A) after his stroke and 6 months thereafter (Figs. 1E and 2E). This pattern of bilateral cerebral activation and cerebellar activation was also present 3.5 years after the onset of his stroke when he was tested with the 3 Tesla magnet (Figs. 1I and 2I).
At Time 1 Patient 2 also showed a pattern of bilateral activation when finger tapping with his right hand (Figs 1B and 2B), but this activity decreased with time. At Time 2, finger tapping with his right hand was associated with activation of the sensorimotor strip and ipsilateral cerebellar hemisphere (Figs. 1F and 2F), a pattern that has been observed in normal individuals. The same pattern was found when he was tested with 3 Tesla magnet (Figs. 1J and 2J).
The differences between finger tapping with the unaffected hand by the two patients were less pronounced. However, they suggest that for Patient 1, greater bilateral activation was associated with finger tapping with both the affected and nonaffected hands and remained so over the 3.5 years of follow-up. In contrast, Patient 2 made a poor recovery with his left hand but maintained good motor recovery with his right hand.

Discussion
In the last few years, numerous studies have correlated changes on fMR imaging with different measures of motor recovery after stroke. Seitz et al.[15] showed that motor recovery after hemiparetic brain infarction was associated with activation patterns far removed from the lesion site. These investigators suggested that their findings supported von Monakow’s concept of diaschisis as the basis of recovery.
Other investigators have argued that the pattern of brain activation observed between “good” and “poor” motor recovery is related to whether the lesion is located subcortically or cortically (and subcortically).[10] Another concept is that the general “intactness” of the impaired corticospinal tract ultimately determines the degree of plasticity.[4]
In an especially provocative study, Ward et al.[17] demonstrated that visually paced hand-grip performance with the affected hand after stroke was correlated with decreased activation over time. That is, better motor performances seemed to be associated with less diffuse patterns of activation and clearly approached the pattern seen in normal individuals performing this task. These investigators, however, noted that increased bilateral brain activation in areas far removed from the sensorimotor regions were associated with recovery in patients who had a lesion in the pons (their Patient 1) and/or lesions of the internal capsule (their Patient 2). This observation was compatible with earlier observations by Weiller et al.[18]
Our present findings support these observations. Ward et al.[17] used a different motor task (visually paced hand-grip task), but their findings suggest that bilateral cerebral activation as well as cerebellum activation may be important in recovery from subcortical lesions as in our Patient and as in Patient 1 in the study by Ward et al.[17] The latter patient was a 53-year-old woman with a lesion in the right pons.
Why is the cerebellum so important for recovery for this type of patient? Ward et al.[17] cited other researchers’ contributions, suggesting that ” . . . the cerebellum is involved in detecting error between internal modes of movement and the sensory consequences of active movement.” Johnson et al.[8] noted that normal preparation for covert movement of either hand is often associated with large regions of activation in the left cerebral hemisphere and right cerebellar hemisphere. Collectively, these findings suggest that the cerebellum may be especially important in initiating and correcting motor movements.
Patients with certain lesions in the internal capsule may have difficulties initiating or performing motor tasks. This may be the basis of hemiparesis in some individuals. Consequently, passive range of movement may be especially helpful in promoting recovery from such lesions.[1]
The role of the cerebellum in initiating and correcting motor function is also consistent with recent interest in constraint-induced therapy after hemiplegia,[16,19] which forces patients to use their affected hand. This type of procedure may be helpful for individuals who have residual neural network capacity to re-initiate or re-experience a normal movement. However, for many patients, the task is quite frustrating, perhaps because their impaired corticospinal tract has minimal residual capacity. Therefore, despite their best efforts, their performance may not always improve.
Unlike mentally preparing for a task or creating a mental image of performing the task, actually performing a motor task like finger tapping may require relative intactness of the cortical-cerebellum interface. As the patient performs the task, a more “efficient” pattern of activation theoretically would occur. Thus, we concur with Ward et al.[17] and Cramer[2] that the “best form” of recovery is associated with a return to the normal brain activation patterns associated with performing a task.
We also hypothesize that individuals who require sustained bilateral and diffuse cerebral activation to perform a simple motor task may experience a greater and more persistent subjective sense of fatigue than those who recover normal activation patterns. Fatigue is a common symptom associated with stroke.[6] To test this hypothesis, both the subjective state of fatigue and actual motor performance could be measured in investigations of the association between patterns of activation on fMR imaging and recovery after stroke.
Conclusions
Recovery of motor function (as measured by speed of finger tapping) after impairment related to a subcortical lesion is a dynamic process that involves both cerebral hemispheres and the ipsilateral cerebellum. This pattern was observed serially using finger tapping with the affected and nonaffected hands of two subjects who had experienced a CVA.
Acknowledgment
Research for this project was made available through the Women’s Board of the Barrow Neurological Foundation.
References
- Brodal A: Self-observations and neuro-anatomical considerations after a stroke. Brain 96:675-694, 1973
- Cramer SC: Functional imaging in stroke recovery. Stroke 35:2695-2698, 2004
- Cramer SC, Bastings EP: Mapping clinically relevant plasticity after stroke. Neuropharmacology 39:842-851, 200
- Feydy A, Carlier R, Roby-Brami A, et al: Longitudinal study of motor recovery after stroke: recruitment and focusing of brain activation. Stroke 33:1610-1617, 2002
- Heaton RK, Grant I, Matthews CG, et al: Comprehensive Norms for an Expanded Halstead- Reitan Battery: Demographic Corrections, Research Findings, and Clinical Applications. Odessa, FL: Psychological Assessment Resources, 1991
- Ingles JL, Eskes GA, Phillips SJ: Fatigue after stroke. Arch Phys Med Rehabil 80:173-178, 1999
- Johnson SC, Prigatano GP: Functional MR imaging during finger tapping. BNI Quarterly 16:37-41, 2000
- Johnson SH, Rotte M, Grafton ST, et al: Selective activation of a parietofrontal circuit during implicitly imagined prehension. Neuroimage 17:1693-1704, 2002
- Lezak MD: Neuropsychological Assessment. New York: Oxford University Press, 1995
- Luft AR, Waller S, Forrester L, et al: Lesion location alters brain activation in chronically impaired stroke survivors. Neuroimage 21:924-935, 2004
- Moore JC: Neuroanatomical considerations relating to recovery of function following brain lesions, in Bach-y-Rita P (ed): Recovery of Function: Theoretical Considerations for Brain Injury Rehabilitation. Toronto, ON: Hans Huber Publishers, 1986, pp 9-91
- Prigatano GP, Wong JL: Speed of finger tapping and goal attainment after unilateral cerebral vascular accident. Arch Phys Med Rehabil 78:847-852, 1997
- Rao SM, Binder JR, Bandettini PA, et al: Functional magnetic resonance imaging of complex human movements. Neurology 43:2311-2318, 1993
- Reitan RM, Wolfson D: Dissociation of motor impairment and higher-level brain deficits in strokes and cerebral neoplasms. Clin Neuropsychol 8:193-208, 1994
- Seitz RJ, Azari NP, Knorr U, et al: The role of diaschisis in stroke recovery. Stroke 30:1844-1850, 1999
- Taub E, Miller NE, Novack TA, et al: Technique to improve chronic motor deficit after stroke. Arch Phys Med Rehabil 74:347-354, 1993
- Ward NS, Brown MM, Thompson AJ, et al: Neural correlates of motor recovery after stroke: a longitudinal fMRI study. Brain 126:2476-2496, 2003
- Weiller C, Ramsay SC, Wise RJ, et al: Individual patterns of functional reorganization in the human cerebral cortex after capsular infarction. Ann Neurol 33:181-189, 1993
- Wolf SL, LeCraw DE, Barton LA, et al: Forced use of hemiplegic upper extremities to reverse the effect of learned nonuse among chronic stroke and head-injured patients. Exp Neurol 104:125-132, 1989
